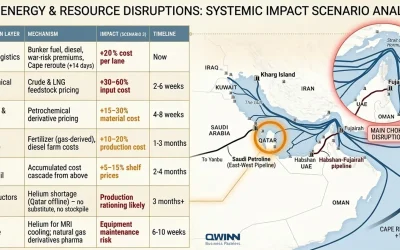
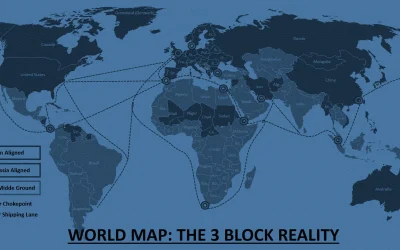
Supply Chain Meets Regulation: What Every Business Leader Needs to Know
A New Reality for Medical Device Supply ChainsAnother EU regulation? Or an opportunity to strengthen your supply chain and gain a competitive edge? Regulation (EU) 2024/1860 introduces new requirements for medical device manufacturers, ensuring that potential supply disruptions are reported well in advance. While this may seem like another compliance challenge, it has real implications for business strategy, risk management, and operational resilience.
For CEOs, CFOs, and COOs, the key questions are simple: What does this regulation mean? How will it impact the business? Should leadership be concerned? And most importantly, how can companies comply without disrupting operations—or even use compliance as a business advantage?
What’s Changing?Regulation (EU) 2024/1860 updates the Medical Device Regulation (MDR) and In Vitro Diagnostic Medical Devices Regulation (IVDR) with Article 10a, a new rule aimed at improving supply chain transparency. Manufacturers must now notify stakeholders at least six months in advance of any planned disruptions. They must also ensure clear communication with distributors, healthcare providers, and regulators, so potential shortages do not come as a surprise.
The goal is simple: reduce sudden shortages of critical medical devices and improve supply chain oversight. But for businesses, this regulation is more than just an extra administrative step—it requires a strategic shift, integrating compliance directly into supply chain planning and operations.
Why It Matters for Business Leaders –
For manufacturers and suppliers, the regulation introduces a new layer of complexity. Companies must now monitor potential disruptions in real-time and take a more structured approach to risk assessment.
This means greater visibility across the supply chain is essential. Businesses must keep track of suppliers, logistics partners, and distributors to ensure they meet reporting requirements. For smaller manufacturers, this can be particularly challenging, as maintaining a robust risk assessment framework and ensuring timely notifications require additional resources.
Failing to comply comes with risks. Regulatory penalties, reputational damage, and operational disruptions could lead to supply shortages, frustrated customers, and increased costs. But for those willing to act early, there is also an opportunity: companies that integrate compliance into their supply chain strategy will benefit from stronger relationships with customers and regulators, greater resilience, and a competitive edge in the market.
How to Comply Without Disrupting BusinessMeeting the requirements of Article 10a does not have to be complex or disruptive. The key is to embed compliance into existing supply chain processes rather than treating it as a separate function.
One of the first steps is to strengthen Integrated Business Planning (IBP) and Sales & Operations Planning (S&OP), ensuring that supply chain risks are identified early. Short-term execution monitoring (S&OE) will also be crucial—companies need to be able to detect potential disruptions quickly and take corrective action before they escalate.
Technology plays an important role in making compliance manageable. Advanced planning systems, digital twin simulations, and early warning tools help organizations track disruptions in real time, ensuring they remain compliant while staying agile. Just as critical is ensuring that Quality Management Systems (QMS) are integrated with supply chain data so that compliance is naturally embedded into day-to-day operations.
Another key element is leadership. QA and RA teams must have a clear role in supply chain decision-making, without creating unnecessary complexity. Encouraging a culture of transparency—where teams feel comfortable raising concerns early—helps ensure that risks are flagged and managed before they become serious issues.
From Compliance to Competitive Advantage – Regulatory changes often require businesses to rethink how they operate, and this is no exception. While compliance may seem like a cost burden at first, companies that take action early can turn it into an advantage. Aligning compliance strategies with supply chain resilience efforts not only reduces operational risks but also improves reliability and builds trust with customers and regulators.
History has shown that companies that prepared early for MDR updates gained a market advantage over those that scrambled to comply at the last minute. The same will be true here—organizations that proactively integrate compliance into their supply chain strategy will be better positioned to lead, while those that delay risk falling behind.
At Qwinn, we help business leaders navigate regulatory complexity while strengthening their supply chain resilience. The choice is clear: react at the last minute or use this as an opportunity to build a stronger, more agile supply chain.
Is your supply chain ready? Let’s make compliance a strategic advantage—connect with Qwinn today.